Launch of "Rapidchip® D-dimer" A Quantitative Assay Kit for Fibrin Degredation Products
December 9, 2013
Sekisui Medical Co., Ltd.
"Rapidchip® D-dimer", a product for the rapid measurement of D-dimer concentration in the diagnosis of thrombotic disease, will be released on the 9th of December by Sekisui Medical (part of the Sekisui Chemical Group; President: Mutsumi Fukuda; Head Office: Tokyo, Chuo Ward).
"Rapidchip® D-dimer", designed exclusively for use on the "Rapidpia®" analyzer, can be used for the diagnosis of Deep Vein Thrombosis (DVT), Pulmonary Thromboembolism (PTE) and the like through the quantitative detection of the useful diagnostic marker D-dimer in either whole blood or plasma, without the need for pre-treatment steps such as dilution. Furthermore, as the system runs on single-use cartridges based on the principal of immunochromotrography, it can be used economically and without loss of reagent, for outpatients or even in emergency situations, with an analysis time of only 10 minutes.
Sekisui Medical has also been distributing "Rapidchip® BNP" since 2009, which makes use of the same analyzer, and with the introduction of "Rapidchip® D-dimer" hopes to create an even more significant contribution to patient care.
Reference Information
- 1Clinical Significance of D-dimer
D-dimer (D-D dimer) is one of the products of the degradation of stable fibrin by plasmin, stable fibrin itself being produced by the crosslinking of fibrin polymers through the action of activated factor XIII in the coagulation/fibrinolysis system. In the blood, D-dimer (DD) exists as one of a number of complexes including YY/DXD, YD/DY, DD/E.
An increase in the level of D-dimer in the blood can indicate ongoing thrombus production and thrombolysis. It is known as an effective marker in the diagnosis of malignant tumors, obstetric conditions, vascular lesions and diseases such as Disseminated Intravascular Coagulation (DIC), Deep Vein Thrombosis (DVT) and Pulmonary Thromboembolism (PTE). - 2Product Overview
Product Name: Rapidchip® D-dimer
Intended Use: Measurement of D-dimer concentration in whole blood or plasma
Assay Principle: Immunochromatographic method using anti-human D-dimer mouse monoclonal antibody.
Assay Range: 0.2 - 15 μg/mL (Citrated whole blood only. Can be diluted 2x using saline to allow for analysis up to 30 μg/mL)
Sample Type: Whole Blood, Plama (Citric Acid, Heparin)
| Product Name | Package Contents | Recommended Retail Price(without tax) |
|---|---|---|
| Rapidchip® D-dimer | 20 Tests (Includes 1 CAL card) | ¥25,000 |
Sold Separately*
| Product Name | Package Contents | Recommended Retail Price(without tax) |
|---|---|---|
| D-dimer Control | 2 conc. x 0.5 mL (1 bottle each) | ¥4,000 |
- *This product is not intended for in-vitro diagnostic use.
- 3Properties
- 1Can use either whole blood or plasma without the need to pre-treat such as dilution.
- 2An assay time of only 10 minute allows for fast results.
- 3A single-use, cartridge type test means no loss of reagent, making it economical.
- 4Product Pictures
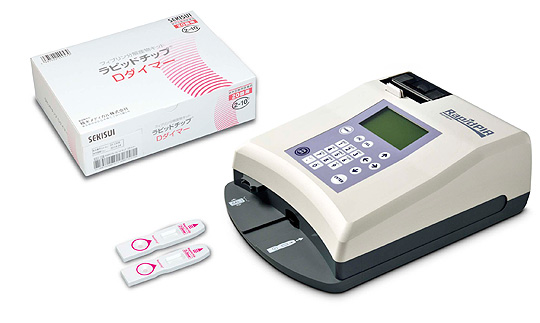
Left: Rapidchip® D-dimer
Right: Rapidpia®Generic Name: Analyser, protein
Medical device requiring specialist maintenance and management
Rapidchip®, Rapidpia® are trademarks owned by Sekisui Medical Co., Ltd. Japan and are registered in Japan and/or other countries.