Coagpia APTT-N
We created this page for medical staff.
- *Read carefully the package insert before use.
Performance Data
Within-run reproducibility
| Sample 1 | Sample 2 | |
|---|---|---|
| 1 | 31.3 | 59.2 |
| 2 | 31.8 | 60.3 |
| 3 | 31.7 | 61.0 |
| 4 | 32.0 | 60.8 |
| 5 | 31.3 | 60.8 |
| 6 | 32.1 | 60.3 |
| 7 | 32.1 | 60.6 |
| 8 | 31.9 | 60.6 |
| 9 | 31.5 | 60.5 |
| 10 | 32.0 | 60.3 |
| Mean | 31.8 | 60.4 |
| S.D. | 0.31 | 0.50 |
| C.V. (%) | 1.00 | 0.83 |
| Max. | 32.1 | 61.0 |
| Min. | 31.3 | 59.2 |
| Range | 0.8 | 1.8 |
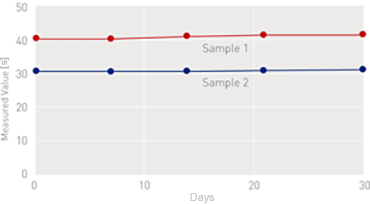
On-Board Stability (After Reconstitution)
[on Coapresta 2000]
Interference
| Concentration | Measured Value(s) | ||
|---|---|---|---|
| Base Plasma | Spiked Plasma | ||
| Unconjugated Bilirubin | 20 mg/dL | 28.0 | 28.5 |
| Conjugated Bilirubin | 20 mg/dL | 28.3 | 28.4 |
| Hemoglobin | 500 mg/dL | 28.5 | 28.2 |
| Chyle | 3000 FTU | 28.3 | 28.4 |
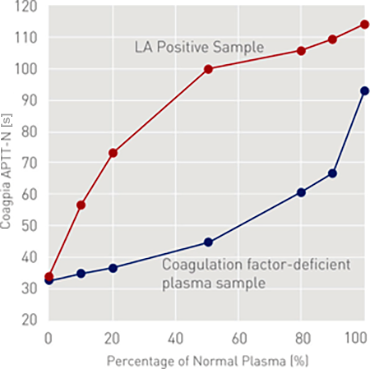
LA Sensitivity (Mixing Study)
Using CP2000's multipoint dilution function, LA positive samples and coagulation factor deficient plasma were measured at 7 points. LA positive sample produces a concave graph while the coagulation factor deficient sample produces a convex graph.
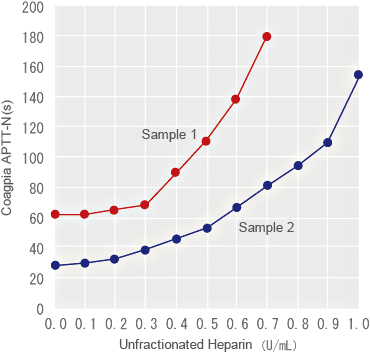
Heparin Sensitivity
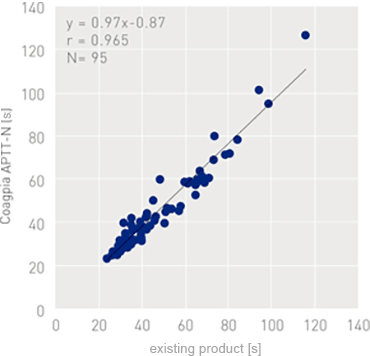
Correlation
Assay Procedure

"Coagpia" is a trademark or registered trademark of SEKISUI MEDICAL CO., LTD. in Japan and/or other countries.
"Coapresta" and all related logos are trademarks or registered trademarks of SEKISUI MEDICAL CO., LTD. in Japan and/or other countries.
We prepared this page for medical staff (doctors, dentists, pharmacists, clinical laboratory technicians, nurses, etc.) in Japan to ensure the correct use of our products. You should be aware that the information provided is not intended for overseas medical staff and the general public.