NORUDIA N HbA1c
We created this page for medical staff.
- *Read carefully the package insert before use.
Features

- Specifically measures the fructosyl dipeptide at the N-terminus of the hemoglobin β-chain
- Applicable to various automated analyzers
- No latex contamination of the cells
- *This package insert follows the Pharmaceuticals, Medical devices and Other Therapeutic Products Act of Japan.
Assay Principle
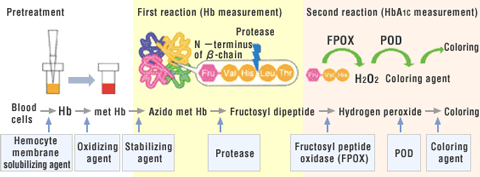
Pretreatment
Red blood cells in the sample are hemolyzed. Hemoglobin is oxidized to form met-hemoglobin.
First Reaction
Fructosyl dipeptide (fructosyl-VH) is cleaved at the N-terminus of hemoglobin β-chain by the action of protease.
Met-hemoglobin is converted into azido met-hemoglobin by the action of sodium azide. Absorbance of the product is measured to determine the hemoglobin concentration.
Second Reaction
Reaction between fructosyl dipeptide (fructosyl-VH) and fructosyl peptide oxidase (FPOX) produces hydrogen peroxide, which reacts with the coloring agent to form color in the presence of peroxidase. Absorbance of the colored substance is measured to determine the HbA1c concentration.
Calculation formula for HbA1c (%)
HbA1c(NGSP %) = 91.5 × HbA1c(μmol/L) / Hb (μmol/L) + 2.15
References : Wieland Hoelzel, Cas Weykamp, et al : Clin. Chem. 50 :166-174, 2004
Package
| Product Name | Package | Storage | |
|---|---|---|---|
| NORUDIA N HbA1c | HbA1c Reagent 1 | 2×30mL | 2-10℃ (Shield from light) |
| HbA1c Reagent 2 | 2×10mL | 2-10℃ | |
| HbA1c Pretreatment Solution for NORUDIA N HbA1c* | 2×200mL | Room Temperature (2-30℃) |
|
| HbA1c Calibrator for NORUDIA N HbA1c* | 3×2conc.×1mL(lyophilized) | 2-10℃ | |
| HbA1c Control for NORUDIA N HbA1c* | 6×2conc.×1mL(lyophilized) | 2-10℃ | |
| HbA1c Blank Solution for NORUDIA N HbA1c* | 1×5mL | 2-10℃ | |
- *Not in vitro diagnostic products in Japan.
Other package sizes are available. Contact SEKISUI MEDICAL CO., LTD. for details.
"NORUDIA" is a trademark or registered trademark of SEKISUI MEDICAL CO., LTD. in Japan and/or other countries.
We prepared this page for medical staff (doctors, dentists, pharmacists, clinical laboratory technicians, nurses, etc.) in Japan to ensure the correct use of our products. You should be aware that the information provided is not intended for overseas medical staff and the general public.